PDF) Consensual guidelines of the Czech National Haemophilia Programme (CNHP) for the dia gnosis and treatment of patients with haemophilia, 3rd edition, year 2021
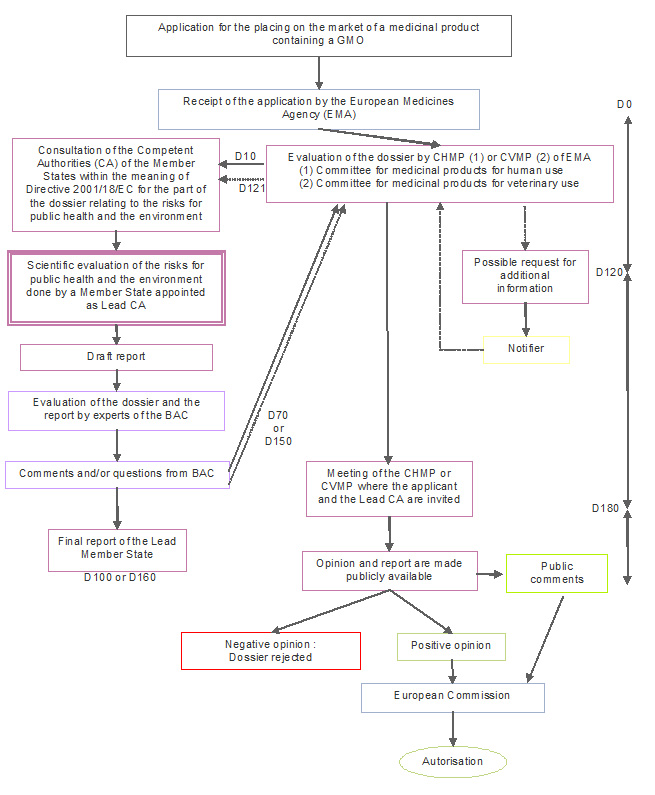
European Medicines Agency procedural advice for users of the centralised procedure for generic/hybrid applications