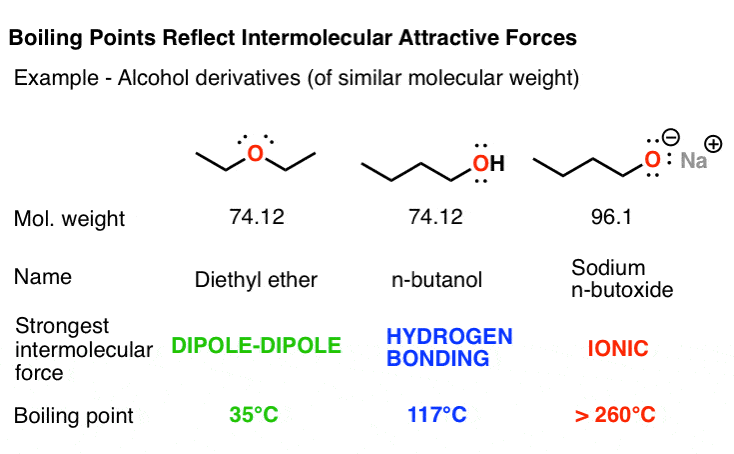
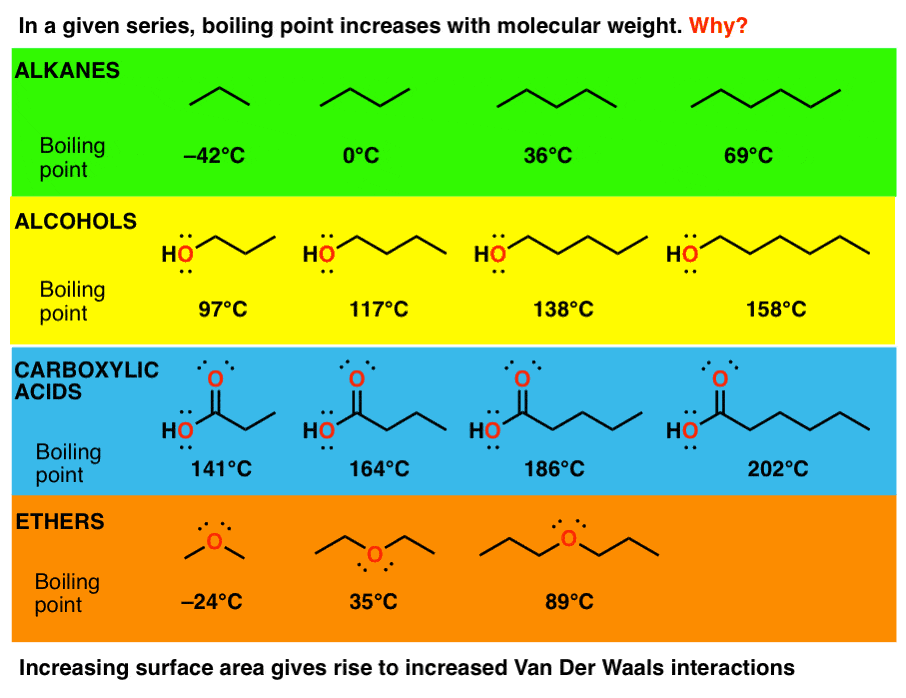
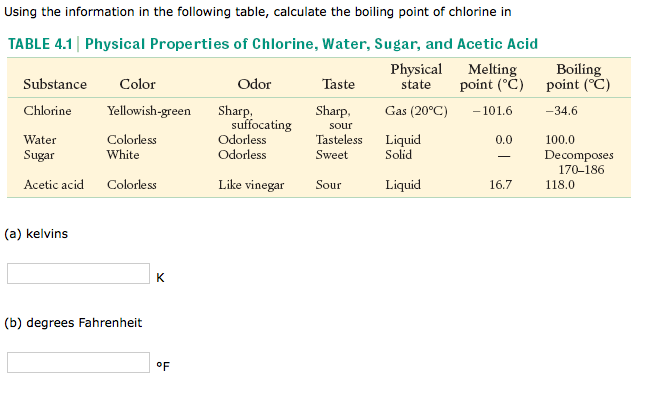
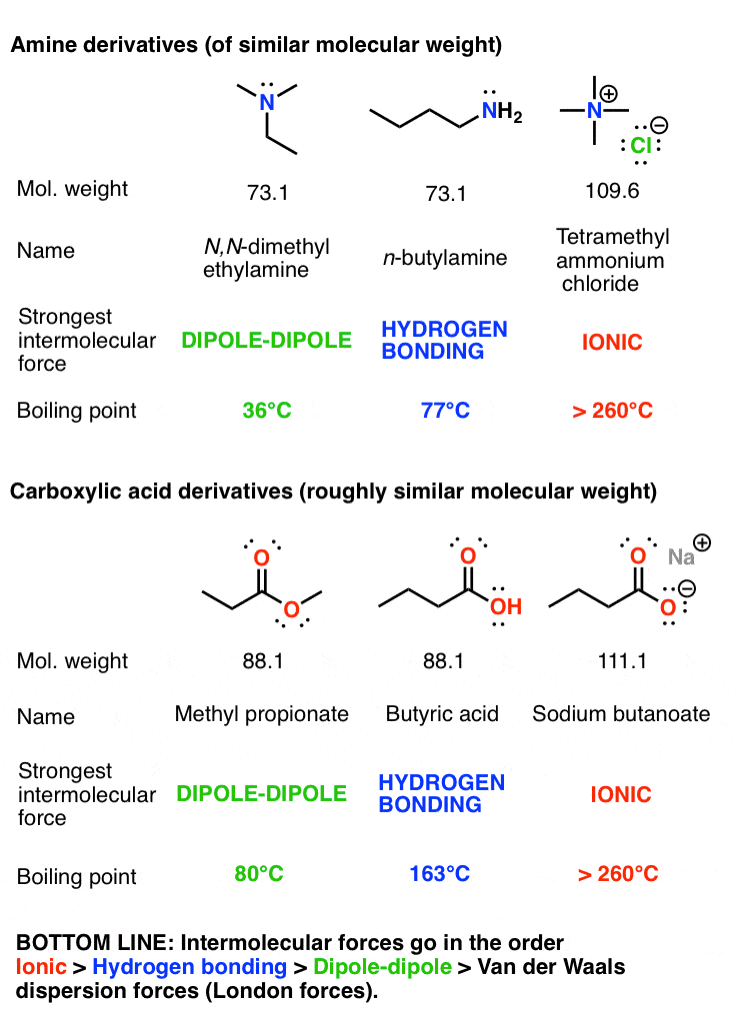
Colligative Properties - Boiling Point Elevation, Freezing Point Depression & Osmotic Pressure - YouTube

SOLVED: Calculate the melting point of benzoic acid (C6H5CO2H), given the following data: ΔΔHfusion = 17.3 kJ/mol and ΔΔSfusion = 43.8 J/(mol K). Select one: a. 0.395 K b. 425 K c. 4250 K d. 395 K

intermolecular forces - How can I determine the highest boiling point given a list of molecules? - Chemistry Stack Exchange

Calculate the mass of ascorbic acid to be dissolved in 75 g acetic acid to lower its melting point by 1.5 ̊C , Kf = 3.9 K kg / mol – The Unconditional Guru